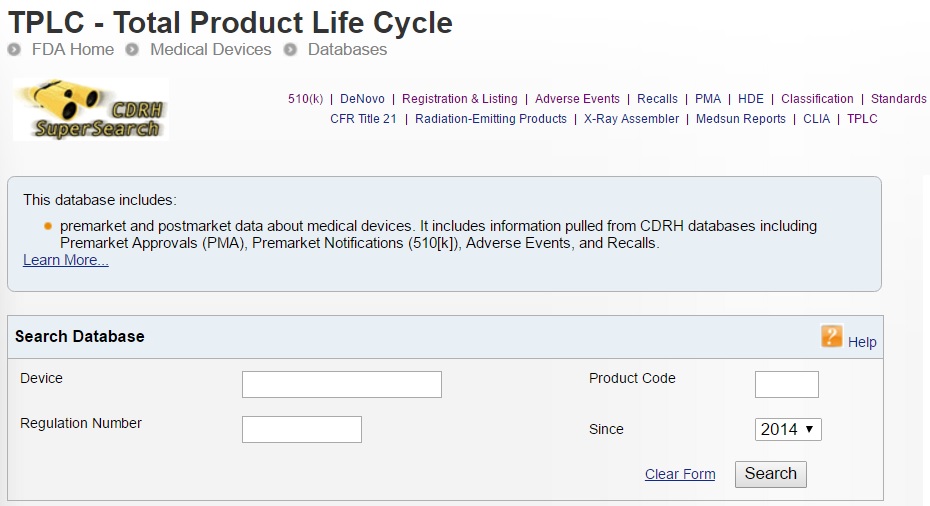
Mhra Adverse Event Medical Device Database . Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web we have launched a new version of the public access registration database (pard). Please visit the new site at: Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web public access database for medical device registrations. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and.
from medicaldeviceacademy.com
Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web we have launched a new version of the public access registration database (pard). Please visit the new site at: Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web public access database for medical device registrations.
Checking adverse event history for your device and competitors
Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Web we have launched a new version of the public access registration database (pard). Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Please visit the new site at: Web public access database for medical device registrations.
From www.medicept.com
Gimme ‘MORE’ U.K.’s MHRA Wants Adverse Events From Device Makers Via Mhra Adverse Event Medical Device Database Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web we have launched a new version of the public access registration database (pard). Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web any event which meets the three reporting criteria. Mhra Adverse Event Medical Device Database.
From learngxp.com
How to Verify Reports of Med Device Adverse Events LearnGxP Mhra Adverse Event Medical Device Database Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web we have launched a new version of the public access registration database (pard). Web public access database for medical device registrations. Please. Mhra Adverse Event Medical Device Database.
From www.slideserve.com
PPT Postmarket Surveillance of Medical Device Adverse Events Mhra Adverse Event Medical Device Database Please visit the new site at: Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web we have launched a new version of the public access registration database (pard). Web any event. Mhra Adverse Event Medical Device Database.
From www.researchgate.net
Medical device Adverse Event reporting form. Download Scientific Diagram Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web report suspected side effects to medicines, vaccines, medical. Mhra Adverse Event Medical Device Database.
From emmainternational.com
FDA Medical Device Databases Backend Platforms Mhra Adverse Event Medical Device Database Please visit the new site at: Web public access database for medical device registrations. Web we have launched a new version of the public access registration database (pard). Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents. Mhra Adverse Event Medical Device Database.
From operonstrategist.com
A Comprehensive Guide to MHRA Medical Device Registration (Steps Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Web public access database for medical device registrations. Please visit the new site at: Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8,. Mhra Adverse Event Medical Device Database.
From www.researchgate.net
Serious adverse events (deaths) reported by the MHRA, EMA and FDA Mhra Adverse Event Medical Device Database Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web public access database for medical device registrations. Web we have launched a new version of the public access registration database (pard). Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident.. Mhra Adverse Event Medical Device Database.
From www.semanticscholar.org
Table 2 from Review of Reported Clinical Information System Adverse Mhra Adverse Event Medical Device Database Please visit the new site at: Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web public access database for medical device registrations. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web we have launched a new version of. Mhra Adverse Event Medical Device Database.
From www.scribd.com
Medical Device Adverse Event Reporting Form Editable PDF Medical Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web public access database for medical device registrations. Web. Mhra Adverse Event Medical Device Database.
From operonstrategist.com
A Comprehensive Guide to MHRA Medical Device Registration (Steps Mhra Adverse Event Medical Device Database Web public access database for medical device registrations. Please visit the new site at: Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web we have launched a new version of. Mhra Adverse Event Medical Device Database.
From www.researchgate.net
Medical device Adverse Event reporting form. Download Scientific Diagram Mhra Adverse Event Medical Device Database Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web public access database for medical device registrations. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Please visit the new site at: Web we have launched a new version of the. Mhra Adverse Event Medical Device Database.
From medicaldeviceacademy.com
Checking adverse event history for your device and competitors Mhra Adverse Event Medical Device Database Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web we have launched. Mhra Adverse Event Medical Device Database.
From knobbemedical.com
MHRA Updates Guidance on Healthcare Apps as Medical Devices Knobbe Mhra Adverse Event Medical Device Database Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8, 5.1.1) is considered an adverse incident. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web we have launched. Mhra Adverse Event Medical Device Database.
From tours.extraview.com
Adverse Event Tracking ExtraView Tours Mhra Adverse Event Medical Device Database Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web public access database for medical device registrations. Web we have launched a new version of the public access registration database (pard). Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web. Mhra Adverse Event Medical Device Database.
From www.greenlight.guru
Medical Device Adverse Event Reporting Regulations EU vs. US Mhra Adverse Event Medical Device Database Please visit the new site at: Web we have launched a new version of the public access registration database (pard). Web public access database for medical device registrations. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8,. Mhra Adverse Event Medical Device Database.
From www.pharmatutor.org
Basics and Essentials of Medical Devices Safety Surveillance PharmaTutor Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Please visit the new site at: Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Web public access database for medical device registrations. Web report suspected side effects to medicines, vaccines, medical device and test kit incidents. Mhra Adverse Event Medical Device Database.
From www.axrem.org.uk
MHRA launches public consultation on future of medical device Mhra Adverse Event Medical Device Database Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Please visit the new site at: Web we have launched a new version of the public access registration database (pard). Web public access database for medical device registrations. Web any event which meets the three reporting criteria (meddev 2.12/1 rev 8,. Mhra Adverse Event Medical Device Database.
From www.pdffiller.com
How To Use The Adverse Event Reporting Form To Document Fill Online Mhra Adverse Event Medical Device Database Web we have launched a new version of the public access registration database (pard). Web report suspected side effects to medicines, vaccines, medical device and test kit incidents used in coronavirus testing and. Web public access database for medical device registrations. Web adverse incidents involving medical devices that occur in the uk must be reported to the medicines and. Please. Mhra Adverse Event Medical Device Database.